Result of the experiment-
I repeated my chosen experiment three times for further accuracy and the results were relatively close to one another. I found that my hypothesis was accurate to the result of my three trials as the powdered tablet took the least amount of time to completely dissolve in the water whereas the tablet which was whole took the most amount of time to dissolve. On average the time for the different sized tablets to dissolve were-
Whole tablet - 1.08 minutes
Half tablet -1.04 minutes
Quarter tablet - 1.03 minutes
Powdered tablet- 16 seconds
Why does a crushed or powdered compound dissolve faster in water than a whole compound?
The reason why the smaller pieces of the tablet did dissolve faster than the larger pieces was because once i had broken them into smaller pieces they took up more surface area in the glass , increasing the surface area of a reactant in this case the Alka-seltzer tablets results in a faster reaction time as shown in my experiment.
The smaller the reactant gets the more surface area it takes up as it is the same amount at the larger pieces except the pieces are much smaller here therefore spreading more. As they were small they took up more space so they reacted much more easily and quickly than they would if they were larger. The powdered tablet did dissolve faster than the whole tablet, because while a powder can contact almost all of the water at once as the tablet at was broken down, a tablet can only contact the water on its surface.
What caused the reaction between the tablet and the water?
The core ingredients in the Alka-Seltzer tablets i used are aspirin, citric acid, and sodium bicarbonate.
Once the sodium bicarbonate dissolved in water, it dissociated meaning it split apart into sodium and bicarbonate ions. The bicarbonate reacts with hydrogen ions from the citric acid to form carbon dioxide and water.
I repeated my chosen experiment three times for further accuracy and the results were relatively close to one another. I found that my hypothesis was accurate to the result of my three trials as the powdered tablet took the least amount of time to completely dissolve in the water whereas the tablet which was whole took the most amount of time to dissolve. On average the time for the different sized tablets to dissolve were-
Whole tablet - 1.08 minutes
Half tablet -1.04 minutes
Quarter tablet - 1.03 minutes
Powdered tablet- 16 seconds
Why does a crushed or powdered compound dissolve faster in water than a whole compound?
The reason why the smaller pieces of the tablet did dissolve faster than the larger pieces was because once i had broken them into smaller pieces they took up more surface area in the glass , increasing the surface area of a reactant in this case the Alka-seltzer tablets results in a faster reaction time as shown in my experiment.
The smaller the reactant gets the more surface area it takes up as it is the same amount at the larger pieces except the pieces are much smaller here therefore spreading more. As they were small they took up more space so they reacted much more easily and quickly than they would if they were larger. The powdered tablet did dissolve faster than the whole tablet, because while a powder can contact almost all of the water at once as the tablet at was broken down, a tablet can only contact the water on its surface.
What caused the reaction between the tablet and the water?
The core ingredients in the Alka-Seltzer tablets i used are aspirin, citric acid, and sodium bicarbonate.
Once the sodium bicarbonate dissolved in water, it dissociated meaning it split apart into sodium and bicarbonate ions. The bicarbonate reacts with hydrogen ions from the citric acid to form carbon dioxide and water.
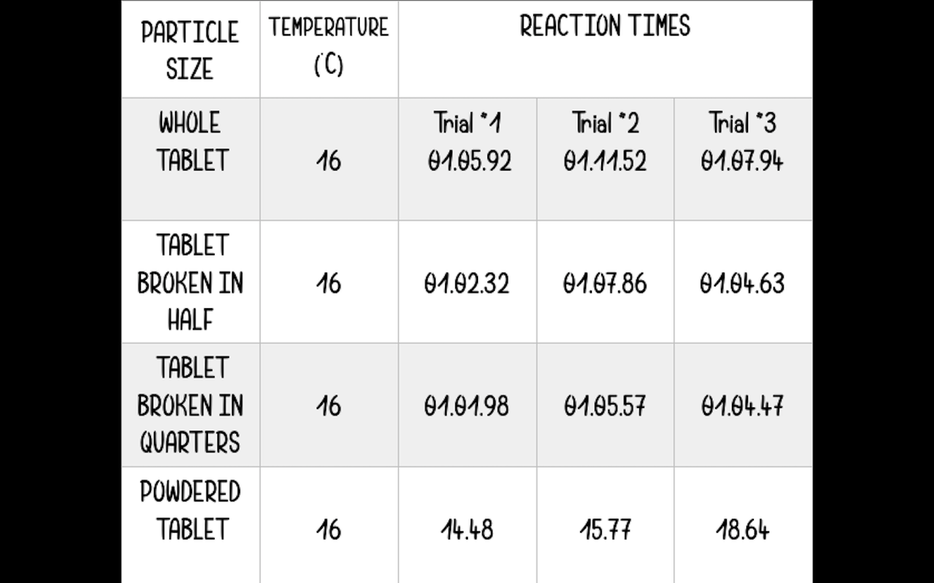
Results in a table-
This shows the temperature of the water at the time the experiment took place and the reactant time for all the three trials i did
As you can see from the information on the table the powdered tablet dissolved the fastest time and as we move up the table and the pieces get larger the time it takes for the tablet to dissolve also increases.
Did the heat of the water matter?
Temperature is related to the time of reaction in my experiment the higher the temperature, the faster the molecules move; the lower the temperature, the slower they move. The temperature of a solution is a measure of its molecules’ average motion and energy.
This shows the temperature of the water at the time the experiment took place and the reactant time for all the three trials i did
As you can see from the information on the table the powdered tablet dissolved the fastest time and as we move up the table and the pieces get larger the time it takes for the tablet to dissolve also increases.
Did the heat of the water matter?
Temperature is related to the time of reaction in my experiment the higher the temperature, the faster the molecules move; the lower the temperature, the slower they move. The temperature of a solution is a measure of its molecules’ average motion and energy.